Call: 07315122850
Inactivated Influenza Vaccine
Price 1200 INR/ Piece
MOQ : 10 Pieces
Inactivated Influenza Vaccine Specification
- Shelf Life
- 2 Years
- Packaging Type
- 1 x 0.5ml
- Medicine Type
- Drug Solutions
- Physical Form
- Liquid
- Quantity/Volume
- 0.5 Milliliter (mL)
- Storage Instructions
- Cool & Dry place
Inactivated Influenza Vaccine Trade Information
- Minimum Order Quantity
- 10 Pieces
- Payment Terms
- Letter of Credit at Sight (Sight L/C)
- Supply Ability
- 1000 Pieces Per Month
- Delivery Time
- 7 Days
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
About Inactivated Influenza Vaccine
Experience transcendent health protection with our exclusive Inactivated Influenza Vaccine. As a heroic solution for influenza prevention, it offers outstanding value and instant savings in safeguarding your health. Every dose contains 0.5 mL of carefully developed vaccine for effective usage, complete with a shelf life up to 15 months if stored appropriately in a dry place. Manufactured with care by leading suppliers in India, this product is available for export worldwide. Achieve unparalleled protection and well-being through this unique preventative measure.
Effective Application of Influenza Vaccine
The Inactivated Influenza Vaccine is suitable for the body, ensuring comprehensive health support against seasonal flu. Its primary application is on human surfaces, specifically administered subcutaneously. Ideal for clinics, hospitals, and vaccination centers, this vaccine provides an outstanding contribution to public health programs. Manufactured in high-grade plants adhering to rigorous standards, it guarantees security against influenza infections like no other.
Wide Market Reach and Supply Capability
The Inactivated Influenza Vaccine is extensively shipped to global markets, with strong export networks ensuring its timely delivery. Orders are systematically processed to meet global demand, emphasizing reliable goods transport logistics. Domestically, its outreach spans across vaccination centers nationwide, ensuring health security everywhere. With ample supply ability, prioritized order fulfillment secures this vaccines availability to meet urgent health requirements worldwide.
Effective Application of Influenza Vaccine
The Inactivated Influenza Vaccine is suitable for the body, ensuring comprehensive health support against seasonal flu. Its primary application is on human surfaces, specifically administered subcutaneously. Ideal for clinics, hospitals, and vaccination centers, this vaccine provides an outstanding contribution to public health programs. Manufactured in high-grade plants adhering to rigorous standards, it guarantees security against influenza infections like no other.
Wide Market Reach and Supply Capability
The Inactivated Influenza Vaccine is extensively shipped to global markets, with strong export networks ensuring its timely delivery. Orders are systematically processed to meet global demand, emphasizing reliable goods transport logistics. Domestically, its outreach spans across vaccination centers nationwide, ensuring health security everywhere. With ample supply ability, prioritized order fulfillment secures this vaccines availability to meet urgent health requirements worldwide.
| Packaging Size | 1 x 0.5ml |
| Brand | Ifluvac Tetra |
| Composition | Inactivated influenza vaccine (NA) |
| Prescription/Non prescription | Prescription |
| Form | Injection |
| Shelf life | 15 Months |
| Pack Size | 0.5 ml |
FAQs of Inactivated Influenza Vaccine:
Q: How is the Inactivated Influenza Vaccine administered?
A: The vaccine is administered subcutaneously at a dosage of 0.5 mL per application.Q: What are the storage instructions for this vaccine?
A: This vaccine should be stored in a dry place to ensure its efficacy and shelf life.Q: When should the vaccine be used for best results?
A: The vaccine is recommended before the flu season begins for optimal preventative protection.Q: Where is this vaccine manufactured and supplied from?
A: The vaccine is manufactured by reputable providers in India and supplied worldwide through systematic export channels.Q: What is the shelf life of the Inactivated Influenza Vaccine?
A: The vaccine has a shelf life of up to 15 months when properly stored in a dry environment.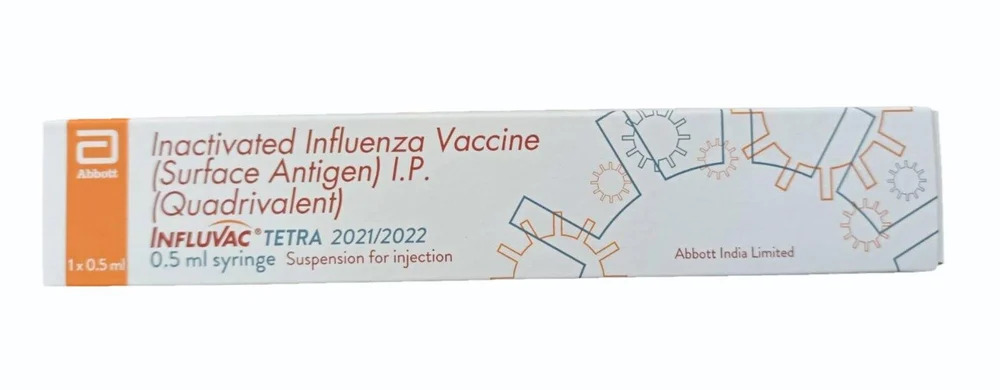
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Vaccines Category
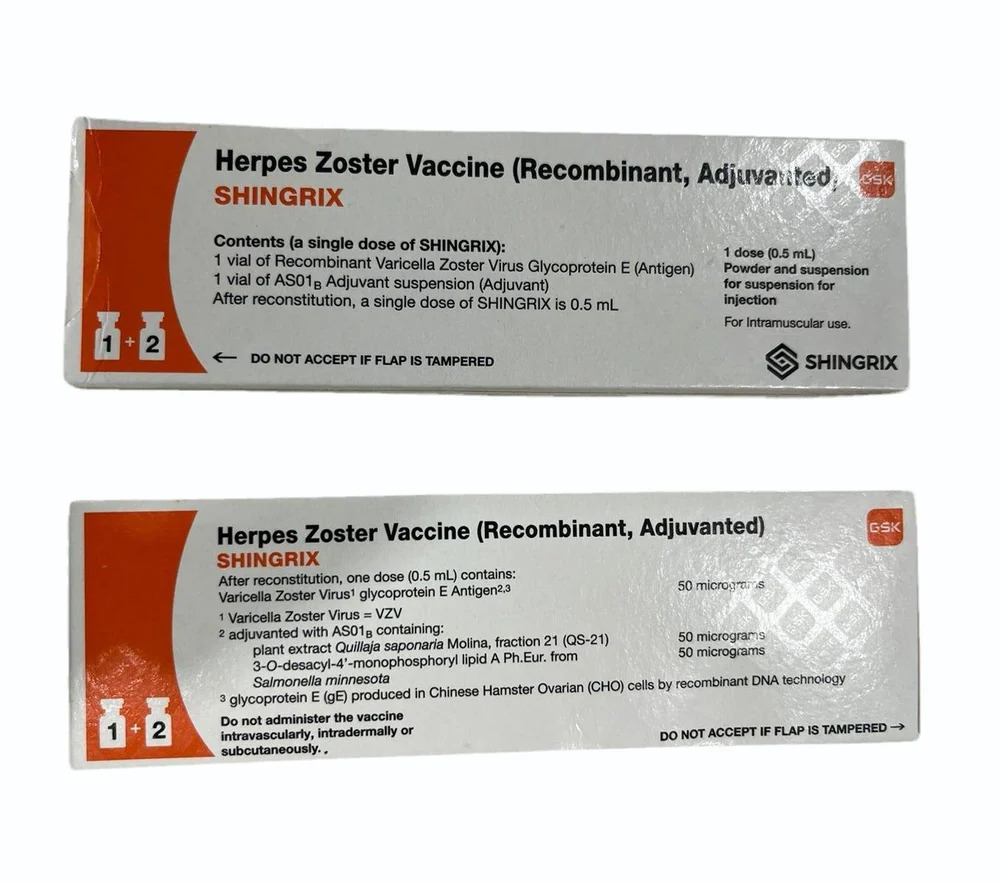
Herpes Zoster Vaccine
Price 8500 INR / Pack
Minimum Order Quantity : 1 Pack
Volume : 0.5 Milliliter (mL)
Age Group : All
Physical Form : Liquid
Medicine Type : Veterinary Injectables

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese